 The elements are stacked in such a way that elements with similar chemical properties form vertical columns, called groups, numbered from 1 to 18 (older periodic tables use a system based on roman numerals). The rows are called periods, and they are numbered from 1 to 7. The elements are arranged in seven horizontal rows, in order of increasing atomic number from left to right and top to bottom. It summarizes huge amounts of information about the elements in a way that facilitates the prediction of many of their properties and chemical reactions. The elements are arranged in a periodic table, which is probably the single most important learning aid in chemistry.  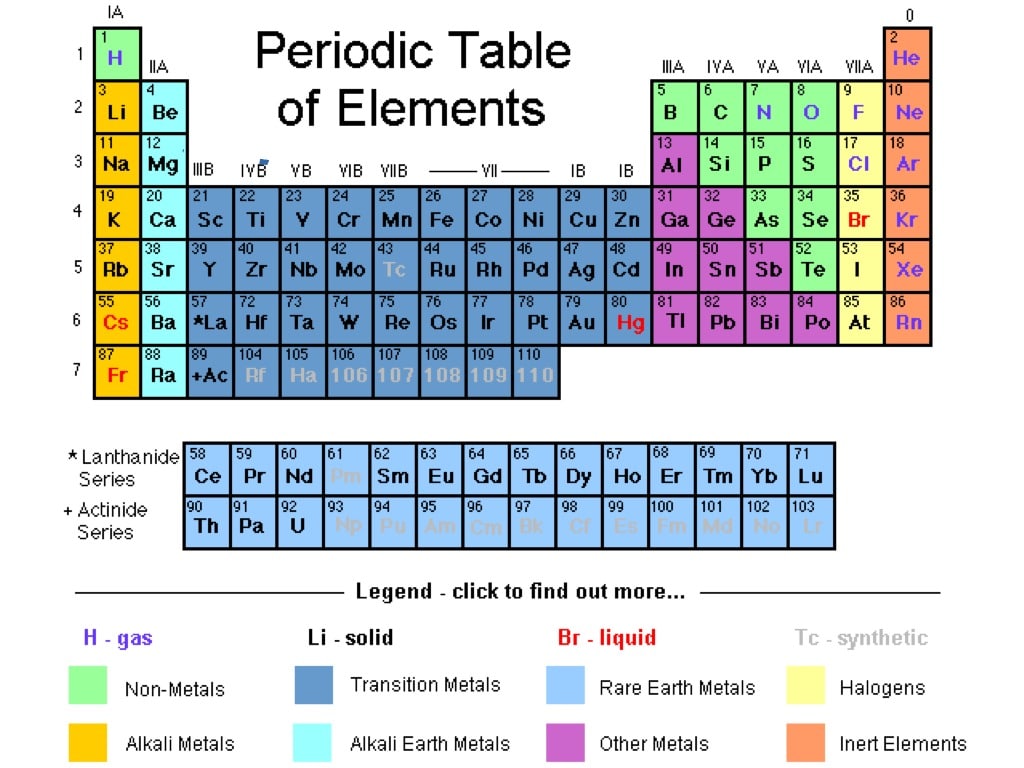
The semimetals lie along a diagonal line separating the metals and nonmetals. The metals are on the bottom left in the periodic table, and the nonmetals are at the top right. In a neutral atom, the number of electrons equals the number of protons.įigure 2.5.1: The Periodic Table Showing the Elements in Order of Increasing Z. The chemistry of each element is determined by its number of protons and electrons. The names of the elements are listed in the periodic table, along with their symbols, atomic numbers, and atomic masses. Each element is assigned a unique one-, two-, or three-letter symbol. The rationale for the peculiar format of the periodic table is explained later. The known elements are arranged in order of increasing Z in the periodic table ( Figure 2.5.1). The atomic number is therefore different for each element. The identity of an element is defined by its atomic number (Z), the number of protons in the nucleus of an atom of the element. Rutherford’s nuclear model of the atom helped explain why atoms of different elements exhibit different chemical behavior. To become familiar with the organization of the periodic table. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
